#30 Quality inspection
Quality inspection
Getting ready for Quality Inspection.
The product should describe the expected physical, chemical, and functional properties of the product together with acceptable measurement limits.
These properties of the product are called Control characteristics.
Common Control characteristics can be length, width, height, diameter, hardness, density, current value, acidity, etc.
Expected values of control characteristics and acceptance limits may vary for each product.
In the product description, you must describe the control characteristics in full detail.
A control characteristic may have different importance in different products. Therefore, the importance classification of characteristics should be done on a product-by-product basis.
The quality inspection sheet is prepared using the control characteristics information in the product descriptions.
Quality Inspection.
Quality control inspection can be categorized as the Final Inspection of finished products and the Incoming Inspection of products supplied through Purchasing.
Both inspections are carried out for product acceptance and are not intended for process control.
The materials you use in your production may be supplied by different suppliers. The quality performance of suppliers will not be the same.
It is necessary to distinguish the successful supplier from the less successful one.
Products and suppliers need to be performance monitored as a pair.
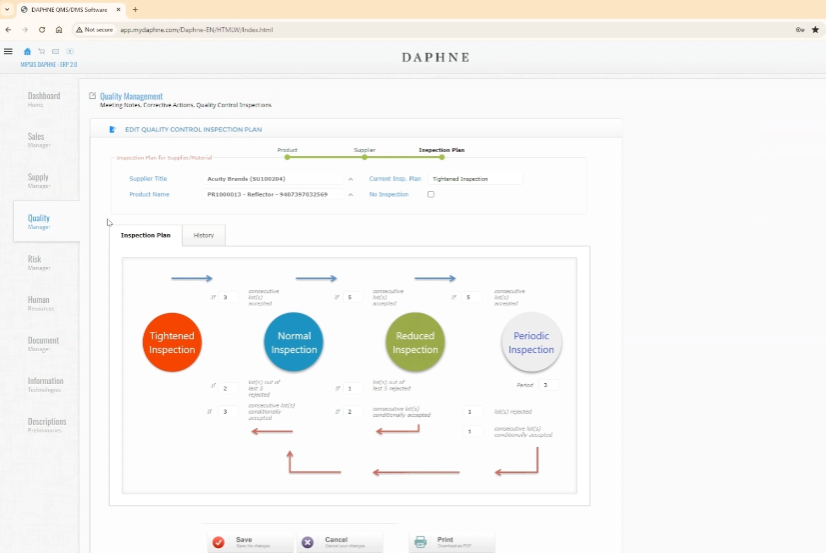
Inspection Plan records of a Supplier-Product pair are opened by the system.
Four inspection plans, Tightened, Normal, Reduced, and Periodic, are applied according to the performance history.
What does changing the Inspection Plan affect? The number of samples taken from lots will change.
Sampling Tables.
Daphne software has nine predefined sampling tables based on the binary combination of Inspection plan and Characteristic importance.
Default plans are considered. Each "inspection plan" and "Importance" pair should have only one default plan.
You can customize the sampling sizes. The Defective column must always be zero. In exceptional cases, it can be used to customize customer needs.
This means that for a batch to be accepted, there must be no defective in the samples.
Quality Inspection Records.
Click the new button on the Inspection Receipts screen. Enter the lot number information required for traceability. There are two options in the Inspection type combo box. These are the Final Inspection and Incoming Inspection options. Select the supplier. Select the warehouse where the product is located for quality control. Enter the product quantity of the lot.
When you press save, the inspection plan and control form are prepared according to the past performance of the supplier-part pair.
The measured values of the characteristics must be entered on the control form.
The form is not mandatory; entering details can sometimes be time-consuming or challenging, so the inspection record can be completed by directly entering the Inspection Result tab.
In addition to acceptance and rejection, conditional acceptance can be selected as a third option. The production plan and time limits for the delivery may require us to accept some of the failed batches.
You must enter the accepted and rejected quantities.
Select the result combo box, save, and complete the process.
